|
1/4/2024 0 Comments Co element ions However, excess of this metal might increase the action of thyroid and bone marrow, which might, in turn, lead to overproduction of erythrocytes, fibrosis in lungs, and asthma. ĭeficiency of cobalt is strongly related to disturbances in vitamin B 12 synthesis, so it might cause anaemia and hypofunction of thyroid and increase the risk of developmental abnormalities and failure in infants. The salts of cobalt stimulate the synthesis of erythropoietin, which is the most important function in the activation of different stages of erythropoiesis, which, in turn, is connected with the formation of erythrocytes in bone marrow. Ĭobalt also plays a role in creating neurotransmitters, which are indispensable for correct functioning of the organism. 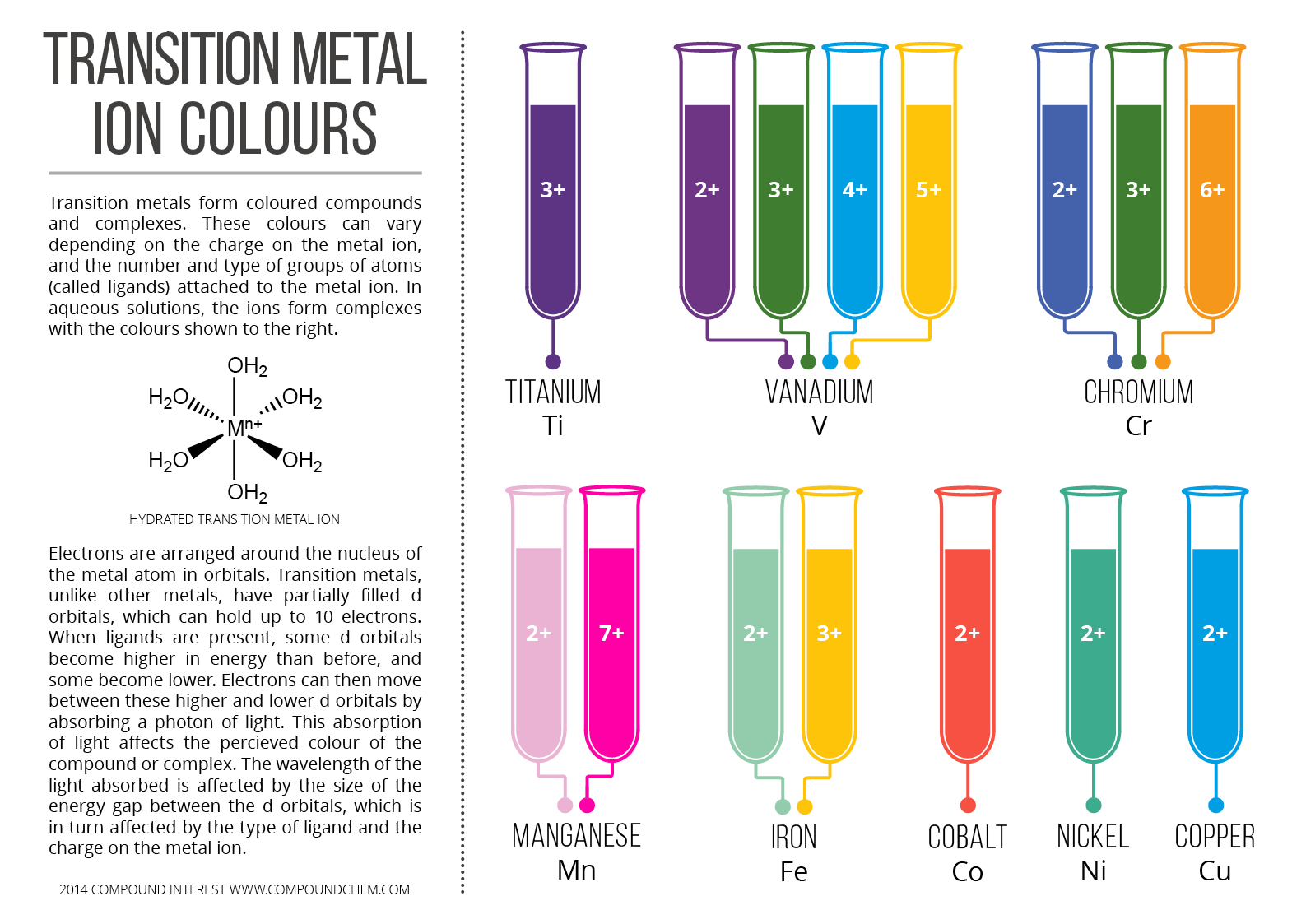
Moreover, cobalt is very important for forming amino acids and some proteins to create myelin sheath in nerve cells. Ĭobalt is a necessary component of vitamin B 12 (hydroxocobalamin) and a fundamental coenzyme of cell mitosis. In the human body this element is present in amounts from 1 to 2 mg: we can find it in the heart, liver, kidney, and spleen, and considerably smaller quantities in the pancreas, brain, and serum. The organic form of cobalt is present in green parts of plants, fish, cereals, and water. 
The first form is essential and necessary for the human body but its excess or deficiency will influence it unfavourably. This essential element occurs in inorganic and organic forms. Thenard's blue, and lacquers in glass and ceramics. Moreover, compounds of cobalt are used in industrial production of paints, e.g. Ĭobalt is used in the production of alloys in plants and in ceramics, as well as chemical catalyses. These two forms produce high-energy γ photons, which can be used in radiotherapy to fight tumours. In nuclear plants, it can occur in two radioactive forms of cobalt, 58Co and 60Co, which are formed from corrosion-erosion of alloys containing cobalt and other metals. This element is used in numerous industrial applications like welding, diamond tooling, grinding, chemical catalyses, and nuclear power plants. It is present in small quantities in compounds with sulphur and arsenic. Ĭobalt is very widespread in the natural environment and can be formed as an effect of anthropogenic activity. Ions form of cobalt, such as Co 3+, react with various acids and create salts however the Co 2+ form is less reactive. The most popular are +2 and +3 other forms of them are rare. Cobalt can occur in various degrees of oxidation forms from +1 to +5. This element belongs to the eighth iron-group. It is one of the essential trace elements in the human body. The ions of cobalt released from the surface of the implant are absorbed by present macrophages, which are involved in many of the processes associated with phagocytose orthopaedic biomaterials particles and release pro-inflammatory mediators such as interleukin-1 (IL-1), interleukin-6 (IL-6), tumour necrosis factor α (TNF-α), and prostaglandin.Ĭobalt was discovered in 1735 by Georg Brandt. 
These released metal ions may cause type IV inflammatory and hypersensitivity reactions, and alternations in bone modelling that lead to aseptic loosening and implant failure. The corrosion of metal is the main problem in the construction of implants. Cobalt and its alloys are fundamental components in orthopaedic implants and have been used for about 40 years. Cobalt gets into the body in several ways: firstly, with food secondly by the respiratory system thirdly, by the skin and finally, as a component of biomaterials. Inorganic forms of cobalt present in ion form, are toxic to the human body, and the longer they are stored in the body, the more changes they cause in cells. Salts of cobalt have been applied in medicine in the treatment of anaemia, as well as in sport as an attractive alternative to traditional blood doping. Its excess or deficiency will influence it unfavourably. The organic form is a necessary component of vitamin B 12 and plays a very important role in forming amino acids and some proteins in nerve cells, and in creating neurotransmitters that are indispensable for correct functioning of the organism. Cobalt is an essential trace element for the human body and can occur in organic and inorganic forms. This element is used in numerous industrial applications and nuclear power plants. Cobalt is widespread in the natural environment and can be formed as an effect of anthropogenic activity. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
